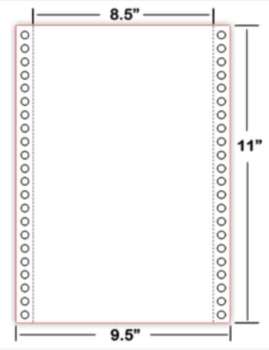
SHOP OUR LATEST PROMOTIONS:
MaxiAids - products designed to promote an Independent, Healthy and Active Lifestyle
Browse a Selection of Categories or View All
Our Customer Favorites
New Products Added... Just For You! Shop All
$22.99
$23.95
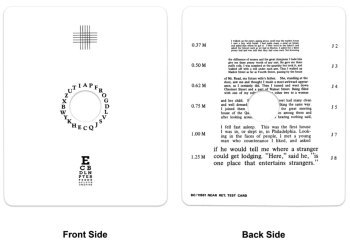
$49.95
$26.95
$14.95
$69.95
$19.95
$6.95
$19.95
$26.95
$19.95
$19.95
$29.95
$26.95
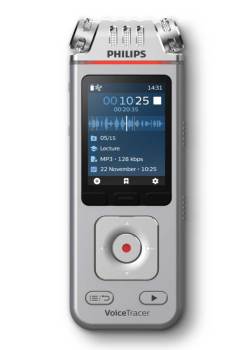
$199.00
$54.95
$7.95
$19.95
$129.99
$19.95
Here's Some Of Our Popular Products:

38 YEARS OF SERVICE
We are here for you

FAST ORDER TURNAROUND
Ship within 1-2 business days

CUSTOMER SERVICE
Only a phone call away ( M-F 8:30- 5:00)